Artificial inflation of apparent photocatalytic activity induced by catalyst-mass-normalization and a method to fairly compare heterojunction systems†
Abstract
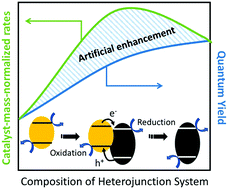
Despite the potential of using photocatalysts to sustainably produce fuels and chemicals, overall reaction rates remain prohibitively low, largely limited by recombination of photogenerated electrons and holes. Past research on suppressing this recombination by the addition of co-catalysts or by the formation of heterojunctions has led to claims that rates show enhancement compared to the single-component, junction-free counterparts. However, comparing these systems is rendered difficult by the common practice of reporting rates on a catalyst mass basis. Using a model system of TiO2 and g-C3N4 with Pt, here we show that normalizing photocatalytic rates by mass fails to provide mechanistic insight and can also artificially inflate the apparent performance of heterojunction systems—in our case, overestimating the enhancement by 24 ± 7%. This effect is caused by the nonlinearity of light absorption with respect to the concentration of each light-absorbing component, according to Beer's law. This factor needs to be taken into account to verify if improvements in absolute activity correspond to intrinsic activity. To this end, a method is presented for approximating a system quantum yield from measured reaction rates under simulated solar or otherwise polychromatic light. A sensitivity analysis provides guidelines for reducing the artificial enhancement introduced by mass-normalization for any heterojunction system.


 Please wait while we load your content...
Please wait while we load your content...