Electronic structure basis for enhanced overall water splitting photocatalysis with aluminum doped SrTiO3 in natural sunlight†
Abstract
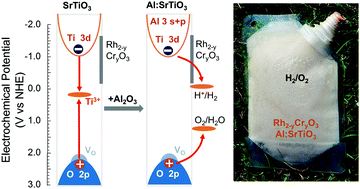
Overall water splitting with photocatalyst particles presents a potentially cost-effective pathway to hydrogen fuel, however, photocatalysts that can compete with the energy conversion efficiency of photovoltaic and photoelectrochemical cells are still lacking. Recently, Goto et al. reported (Joule, 2018, 2(3), 509–520) that Al-doped SrTiO3 microparticles, followed by modification with Rh2−yCryO3 support overall water splitting with 0.4% solar to hydrogen efficiency and with 56% apparent quantum yield at 365 nm. Earlier, based on transient IR spectroscopy results, the improved activity of Al:SrTiO3 had been attributed to the removal of Ti3+ deep recombination sites by the Al3+ ions. Here we use X-ray photoelectron spectroscopy to show that Al3+ incorporation not only reduces the Ti3+ concentration but also diminishes the n-type character of SrTiO3 and shifts the Fermi level to more oxidizing potentials. According to DFT, the electronic structure of Al-doped SrTiO3 depends sensitively on the relative locations of Al3+ and oxygen vacancies sites, with Al3+ ions next to the oxygen vacancies being most effective at suppressing the sub-band gap states. Reduced hole and electron trapping resulting from the elimination of Ti3+ states is confirmed by surface photovoltage spectroscopy and electrochemical scans. These findings not only provide an experimental basis for the superior water splitting activity of Al-doped SrTiO3, under ultraviolet and solar irradiation, but they also suggest that aliovalent doping may be a general method to improve the solar energy conversion properties of metal oxides. Additionally, overall water splitting with a type 1 single bed particle suspension ‘baggie’ reactor under direct sunlight illumination with 0.11% solar to hydrogen efficiency is also demonstrated for the first time. This provides a proof of concept for one of the models in the 2009 US Department of Energy Technoeconomic analysis for photoelectrochemical hydrogen production.
- This article is part of the themed collection: Celebrating Latin American Talent in Chemistry


 Please wait while we load your content...
Please wait while we load your content...