Solvating power series of electrolyte solvents for lithium batteries†
Abstract
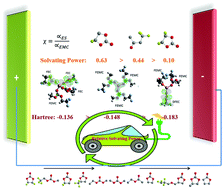
From dictating the redox potential of electrolyte solvents to shaping the stability of solid-electrolyte interfaces, solvation plays a critical role in the electrochemistry of electrolytes. To efficiently design functional electrolytes for lithium batteries, it is particularly important to understand the relative solvating ability of each individual organic solvent, because most of the electrolyte systems are comprised of two or more electrolyte solvents. Using a newly developed internally referenced diffusion-ordered spectroscopy technique and diffusion coefficient–coordination ratio (D–α) analysis, we successfully constructed a solvating power series for common electrolyte solvents. We demonstrated the usefulness of this solvating power series in designing more reliable electrolyte system by selecting an appropriate fluorinated electrolyte solvent for a high-voltage lithium metal battery (LMB) as an example. For a methyl(2,2,2-trifluoroethyl)carbonate-based electrolyte, we identified fluoroethylene carbonate as a more desirable cyclic carbonate co-solvent than difluoroethylene carbonate for LMB due to its significantly higher ability to solvate lithium ions.


 Please wait while we load your content...
Please wait while we load your content...