High-performance oxygen evolution electrocatalysis by boronized metal sheets with self-functionalized surfaces†
Abstract
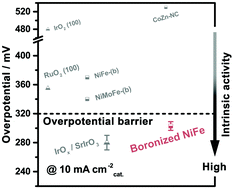
The oxygen evolution reaction (OER) is a key half-reaction involved in many important electrochemical reactions, but this process is quite sluggish and the materials needed usually show unsatisfactory activity, stability or corrosion resistance in the harsh electrocatalysis environment. Here we report an effective boronization strategy for the value-added transformation of inexpensive, commercially available metal sheets (Ni, Co, Fe, NiFe alloys and steel sheets) into highly active and stable, corrosion resistant oxygen evolution electrodes. The boronized metal sheets exhibit OER activities that are an order of magnitude higher than those of the corresponding metal sheets, and show significantly improved catalytic stability and corrosion resistance in the operating environment. The in situ formed, ultrathin (2–5 nm), metaborate-containing oxyhydroxide thin films on metal boride surfaces are identified as a self-functionalized, highly active catalytic phase for the OER. In particular, a boronized NiFe alloy sheet is demonstrated to exhibit intrinsic catalytic activity higher than those of the state-of-the-art materials in 1 M KOH, while retaining such catalytic activity for over 3000 hours. Additionally, the boronized NiFe alloy and steel sheets are also demonstrated to have good catalytic activity as well as excellent catalytic stability and corrosion resistance in 30% KOH solution, a widely-adopted electrolyte in commercial water–alkali electrolyzers.


 Please wait while we load your content...
Please wait while we load your content...