Syntheses, structures and catalytic activity of tetranuclear Mg complexes in the ROP of cyclic esters under industrially relevant conditions†‡
Abstract
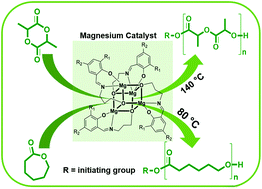
Four tetranuclear magnesium complexes were synthesized and fully characterized. They are excellent catalysts for the ring-opening polymerization (ROP) of bulk racemic-lactide (rac-LA) and ε-caprolactone (ε-CL) even with low catalyst loading under industrially relevant conditions without additional co-initiators, yielding polymers with high molecular weight (Mn) and moderately controlled molecular weight distribution (MWD). The polymerization activity depends on the steric and electronic properties of the imino(phenolate) ligands. Kinetic studies of the most active catalyst 1 confirmed that the ROP of rac-LA and ε-CL has a first order dependence on monomer concentration. The ROP of rac-LA and ε-CL using 1–4 with BnOH gave polymers with narrow MWD and a close correlation between Mobsn and Mtheon values. Catalyst 1 was also capable of polymerizing technical-grade rac-LA and ε-CL even with large monomer-to-catalyst ratios ([M]0/[C]0) of up to 10 000 : 1 and without any additional co-initiator. The very high turnover frequencies (TOF) of 9600 h−1 (rac-LA) and 24 000 h−1 (ε-CL) prove that 1 belongs to the most active biocompatible, sustainable and nontoxic ROP catalysts under industrially relevant conditions.


 Please wait while we load your content...
Please wait while we load your content...