Bis(alkyl) scandium and yttrium complexes coordinated by an amidopyridinate ligand: synthesis, characterization and catalytic performance in isoprene polymerization, hydroelementation and carbon dioxide hydrosilylation†
Abstract
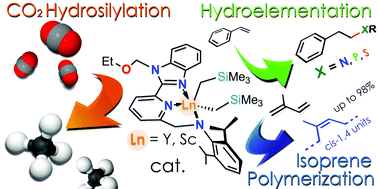
New neutral bis(alkyl) Sc and Y complexes [N,Npy,N−]Ln(CH2SiMe3)2(THF)n [n = 0, Ln = Sc (1Sc), Y (1Y); n = 1, Ln = Y (1YTHF)] stabilized by a tridentate monoanionic amidopyridinate ligand were straightforwardly prepared by alkane elimination, upon mixing ligand [N,Npy,N−]H and metal precursor Ln(CH2SiMe3)3(THF)2 in toluene at 0 °C. Depending on the work-up conditions, yttrium bis(alkyl)s were isolated as either a pentacoordinate Lewis base free complex [N,Npy,N−]Y(CH2SiMe3)2 (1Y) or as a hexacoordinate THF adduct [N,Npy,N−]Y(CH2SiMe3)2THF (1YTHF). For the smaller Sc ion the only solvent-free complex [N,Npy,N−]Y(CH2SiMe3)2 (1Sc) was isolated as a pentacoordinate species irrespective of the reaction/work-up/crystallization conditions applied. Complexes 1Ln (Ln = Y, Sc) and 1YTHF were scrutinized as pre-catalysts in ternary catalytic systems Ln/borate/AliBu3 (borate = [HNMe2Ph][B(C6F5)4] or [Ph3C][B(C6F5)4]), applied to isoprene (IP) polymerization, providing moderate activity albeit high selectivity with predominant formation of 1,4-cis polyisoprene (up to 99%). The same complexes proved to be effcient catalysts also for the intermolecular hydrolelementation of styrene with various EH sustrates (pyrrolidine, morpholine, Ph2PH, PhPH2, PhSH) affording linear anti-Markovnikov addition products exclusively. After a preliminary activation by B(C6F5)3, selected bis(alkyl) complexes from this series have been finally used as valuable pre-catalysts for the CO2 hydrosylilation to CH4 in the presence of organosilanes as reducing agents (PhMe2SiH, PhSiH3, Et2MeSiH).


 Please wait while we load your content...
Please wait while we load your content...