Filling the void: controlled donor–acceptor interaction facilitates the formation of an M–M single bond in the zero oxidation state of M (M = Zn, Cd, Hg)†
Abstract
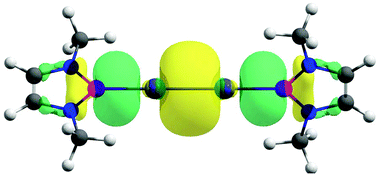
The intriguing question of whether it is possible to form a genuine M0–M0 single bond for the M2 species (M = Zn, Cd, Hg) is addressed here. So far, all the bonds reported in the literature are exclusively MI–MI. Herein, we present viable M2(NHBMe)2 (M = Zn, Cd, Hg; NHBMe = (HCNMe)2B) complexes in which the controlled donor–acceptor interaction leads to an M0–M0 single bond. In these complexes, M2 in the 1∑g ground state with the (nσg+)2(nσu+)2 (n = 7, 10 and 14 for M = Zn, Cd and Hg, respectively) valence electron configuration forms donor–acceptor bonding with singlet 2NHBMe ligands where a combined effect of dominant (+,−) σ-backdonation from the antibonding (nσu+)2 orbital of M2 to the 2NHBMe ligands and a somewhat weaker (+,+) σ-donation from the 2NHBMe ligands to the bonding (n + 1)σg+ orbital leads to the unorthodox bonding situation of forming an M–M single bond in the zero oxidation state by eventually nullifying one effect by another. This is an unprecedented situation in the sense that the NHBMe ligand acts as a strong σ-acceptor and a weaker σ-donor. A comparison with the experimentally reported M2(PhDipp)2 complexes reveals the uniqueness of the NHBMe ligand in exhibiting such a bonding scenario. The M2(NHBMe)2 complex is thermochemically viable with respect to possible dissociation channels at room temperature, except for metal extrusion processes, M2(NHBMe)2 → M + M(NHBMe)2 and M2(NHBMe)2 → M2 + (NHBMe)2. Although the latter two processes are exergonic, they are kinetically protected by a high free energy barrier of 26.5–39.5 kcal mol−1. The experimental characterization of M2(PhDipp)2 despite similar exergonic channels reveals such kinetic stability to be enough for the viability of the M2(NHBMe)2 complexes. Furthermore, the ligand exchange reaction considering M2(PhMe)2 as the starting material also turned out to be feasible. Therefore, the M2(NHBMe)2 complexes are the first cases that feature a neutral M2 moiety with a single M0–M0 covalent bond, where M is a Group 12 metal.


 Please wait while we load your content...
Please wait while we load your content...