Hydrogenation and N-alkylation of anilines and imines via transfer hydrogenation with homogeneous nickel compounds†
Abstract
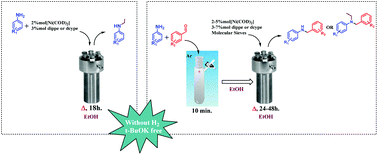
The nickel-catalyzed N-alkylation of a variety of arylamines via transfer hydrogenation in the absence of pressurized hydrogen and basic or acidic additives was achieved in a tandem reaction. This process was further extended to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond reduction and N-alkylation of a variety of imines with ethanol, the latter acting as a hydrogen and acetaldehyde source, which allowed for the reduction and subsequent condensation to yield the corresponding N-alkylated products.
N bond reduction and N-alkylation of a variety of imines with ethanol, the latter acting as a hydrogen and acetaldehyde source, which allowed for the reduction and subsequent condensation to yield the corresponding N-alkylated products.
- This article is part of the themed collection: Celebrating recent chemical science in Mexico


 Please wait while we load your content...
Please wait while we load your content...