Molecular squares, coordination polymers and mononuclear complexes supported by 2,4-dipyrazolyl-6H-1,3,5-triazine and 4,6-dipyrazolylpyrimidine ligands†‡
Abstract
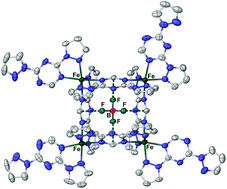
The Fe[BF4]2 complex of 2,4-di(pyrazol-1-yl)-6H-1,3,5-triazine (L1) is a high-spin molecular square, [{Fe(L1)}4(μ-L1)4][BF4]8, whose crystals also contain the unusual HPzBF3 (HPz = pyrazole) adduct. Three other 2,4-di(pyrazol-1-yl)-6H-1,3,5-triazine derivatives with different pyrazole substituents (L2–L4) are unstable in the presence of first row transition ions, but form mononuclear, polymeric or molecular square complexes with silver(I). Most of these compounds involve bis-bidentate di(pyrazolyl)triazine coordination, which is unusual for that class of ligand, and the molecular squares encapsulate one or two BF4−, ClO4− or SbF6− ions through combinations of anion⋯π, Ag⋯X and/or C–H⋯X (X = O or F) interactions. Treatment of Fe[NCS]2 or Fe[NCSe]2 with 4,6-di(pyrazol-1-yl)-2H-pyrimidine (L5) or its 2-methyl and 2-amino derivatives (L6 and L7) yields mononuclear [Fe(NCE)2L2] and/or the 1D coordination polymers catena-[Fe(NCE)2(μ-L)] (E = S or Se, L = L5–L7). Alcohol solvates of isomorphous [Fe(NCS)2L2] and [Fe(NCSe)2L2] compounds show different patterns of intermolecular hydrogen bonding, reflecting the acceptor properties of the anion ligands. These iron compounds are all high-spin, although annealing solvated crystals of [Fe(NCSe)2(L5)2] affords a new phase exhibiting an abrupt, low-temperature spin transition. Catena-[Fe(H2O)2(μ-L5)][ClO4]2 is a coordination polymer of alternating cis and trans iron centres.


 Please wait while we load your content...
Please wait while we load your content...