Structural variability of pendant groups within the interlayer region of zirconium arene-phosph(on)ates: chemical and structural characterization of oxy- and methyl-linked 2-naphthyl phosphonates, and mixed oxy-linked derivatives†
Abstract
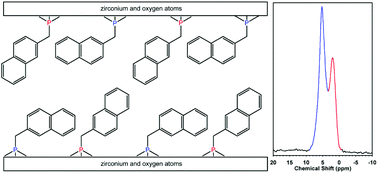
Several new zirconium phosphonates incorporating the naphthalene ring and having the general formula Zr(O3PR)1(O3PR′)1 [R and R′ = –CH2C10H7, –OC10H7, –CH3, –OC2H5, –OH] have been synthesized. These materials were chemically characterized using thermal gravimetric analysis (percentage of organic content), infrared spectroscopy (presence of the desired organic functional groups), and solid-state 31P NMR (phosphorus environments), while the structural parameters were determined using X-ray powder diffraction (interlayer d spacings). The two new zirconium bis(phosphonates), Zr(O3PC10H7)2 and Zr(O3PCH2C10H7)2, were found to have d spacings of 19.6 and 20.0 Å, respectively. Three of the four zirconium mixed phosphonates examined are found to be single-phase structures with random mixtures of the organic moieties within the interlayer, and possess d spacings (14.3, 15.3, and 16.1 Å) that are between those of the two parent zirconium bis(phosphonates). The fourth is found to be a staged or segregated structure and possesses a d spacing that is approximately a sum of the two parent zirconium bis(phosphonates), with a d spacing of 28.2 Å. Solid-state 31P NMR of Zr(O3PCH2C10H7)2 revealed the presence of two isotropic resonances, which is interpreted in terms of two distinct, “locked-in” conformations of the –CH2C10H7 group. The experimental d spacings of the zirconium bis(phosphonates) correlate well with a simple predictive model based on the effective length and predominant conformation of the organic functional group.
- This article is part of the themed collection: Inorganic Porous and Layered Material


 Please wait while we load your content...
Please wait while we load your content...