Superfine CoNi alloy embedded in Al2O3 nanosheets for efficient tandem catalytic reduction of nitroaromatic compounds by ammonia borane†
Abstract
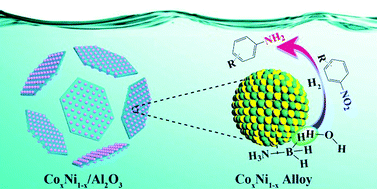
Aromatic amino compounds are important and universally used chemical intermediates in a wide range of industrial fields. Thus, their production with high efficiency and selectivity under ambient conditions is expected and demanded in modern industry. Herein, a series of superfine CoNi alloy nanoparticles embedded in Al2O3 nanosheet (CoxNi1−x/Al2O3, where x represents the content of Co in the precursor) catalysts was fabricated from CoNiAl-LDH and used to catalyze the tandem dehydrogenation of ammonia borane (AB) and hydrogenation of nitroaromatics to the corresponding amines. Systematic experiments indicate that the composition, size, morphology and catalytic performance of the CoxNi1−x/Al2O3 catalysts can be easily controlled by changing the content of Ni in the CoNiAl-LDH precursor. Particularly, Co0.67Ni0.33/Al2O3 exhibited the best tandem catalytic performance among the six samples. This as-prepared catalyst not only showed a moderate turn-over-frequency value (TOF: 34.5 molH2 molCo0.67Ni0.33−1 min−1 at 298 K without base or additives) and relatively low activation energy (32.4 kJ mol−1) for the dehydrogenation of AB, but also superior catalytic activity (conversion yield reaching up to 100%) and selectivity (>99%) for the tandem reductive transformation of in excess of sixteen types of nitroaromatics to aromatic amines. Density functional theory (DFT) calculations suggest that the construction of the CoNi alloy optimized the electronic structure with respect to the pure component, promoting its activity for AB hydrolysis and nitroaromatics hydrogenation. Finally, the catalyst could be easily recycled using a magnet due to the magnetic properties of the Co0.67Ni0.33 alloy.


 Please wait while we load your content...
Please wait while we load your content...