Low-temperature synthesis of superconducting iron selenide using a triphenylphosphine flux†
Abstract
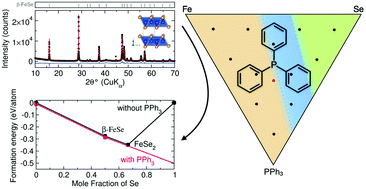
Many functional materials have relatively low decomposition temperatures (T ≤ 400 °C), which makes their synthesis challenging using conventional high-temperature solid-state chemistry. Therefore, non-conventional techniques such as metathesis, hydrothermal, and solution chemistry are often employed to access low-temperature phases; the discovery of new chemistries is needed to expand access to these phases. This contribution discusses the use of triphenylphosphine (PPh3) as a molten flux to synthesize superconducting iron selenide (Fe1+δSe) at low temperature (T = 325 °C). Powder X-ray diffraction and magnetism measurements confirm the successful formation of superconducting iron selenide while nuclear magnetic resonance spectroscopy and in situ X-ray diffraction show that the formation of superconducting FeSe at low temperatures is enabled by an adduct between the triphenylphosphine and selenium. Exploration of the Fe–Se–PPh3 phase space indicates that the PPh3–Se adduct effectively reduces the chemical potential of the selenium at high concentrations of triphenylphosphine. This contribution demonstrates that the use of a poorly-solvating yet reactive flux has the potential to enable the synthesis of new low-temperature phases of solid materials.


 Please wait while we load your content...
Please wait while we load your content...