Tailored protective groups for surface immobilization of ruthenium dyes†
Abstract
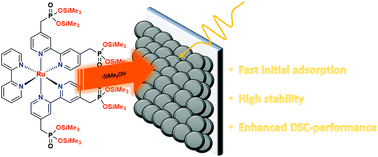
McKenna reaction conditions are applied to the [Ru(4,4′-(CH2PO3Et2)2(bpy)](PF6)2 model chromophore in order to obtain [Ru(4,4′-(CH2PO3TMS2)2(bpy)](Br2−x)(PF6)x (x = 0–2) (2) by replacing the alkyl moieties of the phosphonates with TMS groups (TMS = trimethylsilyl). The model complex is immobilized onto both NiO powder and NiO electrodes on FTO (fluorine doped tin oxide) using organic solvents. The stability of surface binding in aqueous media and the DSC performance of 2 are tested and compared to those of a conventional dye of structure [Ru(4,4′-(CH2PO3TBA2)2(bpy)](PF6)2 (1) (TBA = tetrabutyl ammonium). This is the first example of a ruthenium based chromophore with a phosphonic acid silyl-ester being directly immobilized onto a NiO surface. In addition, complex 2 exhibits superior stability towards desorption in aqueous media and at the same time showing improved DSC performance and stability in acetonitrile and a slightly higher dye loading on the electrode surface compared to 1.


 Please wait while we load your content...
Please wait while we load your content...