Efflorescence on calcareous objects in museums: crystallisation, phase characterisation and crystal structures of calcium acetate formate phases†‡
Abstract
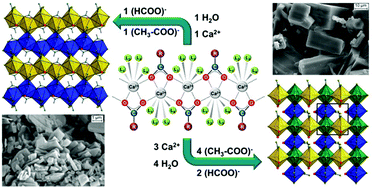
During the systematic investigation of the ternary system Ca(CH3COO)2–Ca(HCOO)2–H2O at room temperature, the congruent crystallisation of two solid calcium acetate formates was observed: the hitherto unknown Ca3(CH3COO)4(HCOO)2·4H2O and the poorly characterised Ca(CH3COO)(HCOO)·H2O. The latter is a frequently observed efflorescence phase found on calcareous objects and it could be also identified as a corrosion phase in a natural history collection of birds’ eggs. Elemental and thermal analyses were employed to determine the phase compositions and by Raman and IR spectroscopy the presence of acetate and formate anions in both solids was confirmed. Laboratory X-ray powder diffraction data were used to solve the crystal structures. Ca3(CH3COO)4(HCOO)2·4H2O crystallises in a primitive tetragonal unit cell with space group P41212 and lattice parameters of a = 6.8655(1) Å and c = 45.5454(6) Å, while Ca(CH3COO)(HCOO)·H2O crystallises in a primitive monoclinic unit cell with space group P21/c and lattice parameters of a = 9.2729(1) Å, b = 6.8002(1) Å, c = 11.2219(2) Å and β = 121.232(1)°. Calcium carboxylate zig-zag chains [Ca(μ2-RCOO)+]n (R = CH3 or H) are the main motif of both crystal structures. In Ca3(CH3COO)4(HCOO)2·4H2O these chains are exclusively composed of acetate anions, whereas in Ca(CH3COO)(HCOO)·H2O only formate anions are situated in the chains. The remaining places in the 7–8 fold coordination sphere of the calcium cations are filled by water molecules and additional carboxylate anions that interconnect neighbouring chains, which eventually leads to layered motifs in both structures.


 Please wait while we load your content...
Please wait while we load your content...