Dioxygen reactivity of iron(ii)–gentisate/1,4-dihydroxy-2-naphthoate complexes of N4 ligands: oxidative coupling of 1,4-dihydroxy-2-naphthoate†
Abstract
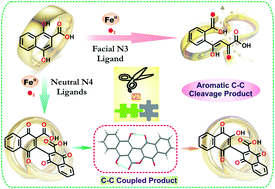
The influence of supporting ligands and co-ligands on the dioxygen reactivity of a series of iron(II) complexes, [(6-Me3-TPA)FeII(GN-H)]+ (1), [(6-Me3-TPA)FeII(DHN-H)]+ (1a), [(BPMEN)FeII(GN-H)]+ (2), [(BPMEN)FeII(DHN-H)]+ (2a), [(TBimA)FeII(GN-H)]+ (3), and [(TBimA)FeII(DHN-H)]+ (3a) (GN-H2 = 2,5-dihydroxybenzoic acid and DHN-H2 = 1,4-dihydroxy-2-naphthoic acid) of N4 ligands, is presented. The iron(II)–gentisate complexes react with dioxygen to afford the corresponding iron(III) species. On the contrary, DHN-H undergoes oxidative C–C coupling to form [2,2′-binaphthalene]-1,1′,4,4′-tetrone 3-hydroxy-3′-carboxylic acid (BNTHC) on 1a, and [2,2′-binaphthalene]-1,1′,4,4′-tetrone 3,3′-dicarboxylic acid (BNTD) on 2a and 3a. In each case, the reaction proceeds through an iron(III)–DHN species. The X-ray single crystal structures of [(6-Me3-TPA)FeII(BNTD)] (1Ox) and [(BPMEN)FeII(BNTD)] (2Ox) confirm the coupling of two DHN-H molecules. The formation of iron(III) product without any coupling of co-ligand from the complexes, [(BPMEN)FeII(HNA)]+ (2b) and [(BPMEN)FeII(5-OMeSA)]+ (2c) (HNA = 1-hydroxy-2-naphthoate, 5-OMeSA = 5-methoxysalicylate) confirms the importance of para-hydroxy group for the coupling reaction. The unusual coupling of DHN-H by the iron(II) complexes of the neutral N4 ligands is distinctly different from the oxygenolytic aromatic C–C cleavage of DHN by the iron(II) complex of a facial N3 ligand.


 Please wait while we load your content...
Please wait while we load your content...