Implications of including a magnetic ion (Cr3+ and Fe3+) at the vanadium site in a geometrically frustrated spinel MgV2O4: magnetic and catalytic properties†
Abstract
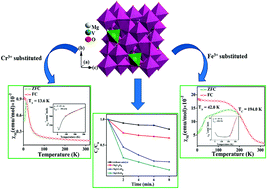
The vanadium sublattice in a geometrically frustrated MgV2O4 is substituted partially with Cr3+ and Fe3+. With a successful Rietveld refinement of the powder X-ray diffraction patterns of MgVCrO4 and MgVFeO4, Cr and Fe occupy the octahedral sites of a spinel structure. Extensive field-dependent and temperature magnetic measurements on these samples reveal exciting results. MgV2O4 remains paramagnetic till 3 K, with a small divergence at around 20 K between ZFC and FC data. MgVCrO4 exhibits antiferromagnetic behavior with a Néel temperature of 13.6 K. MgVFeO4 shows spin-glass behavior resulting from the frustration with a glass transition temperature of 194 K. This sample shows a typical ferromagnetic behavior, with a coercivity of 194.5 Oe within an applied field of ±2 kOe. All three systems have been found to have a frustration index in the range of 1–25, and the effective magnetic moment decreases in the order MgVFeO4 > MgVCrO4 > MgV2O4. While the inclusion of chromium does not alter the bandgap of MgV2O4, iron substitution increases the bandgap. The partial replacement of V3+ with Cr3+ and Fe3+ increases the catalytic ability of the system in terms of the oxidative degradation of methylene blue dye. The catalytic efficiency follows the order MgVFeO4 > MgVCrO4 > MgV2O4, matching well with the trend noticed in the porosity, surface area, and redox ability of Fe3+, Cr3+ and V3+ in these samples. The degradation pathway has been followed by analyzing the intermediates from these experiments by mass spectrometry, and a plausible mechanism is proposed.


 Please wait while we load your content...
Please wait while we load your content...