Nickel complexes of ligands derived from (o-hydroxyphenyl) dichalcogenide: delocalised redox states of nickel and o-chalcogenophenolate ligands †
Abstract
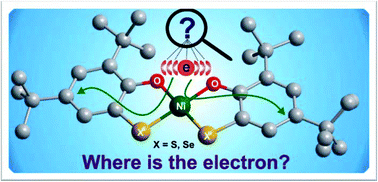
Two monoanionic nickel complexes Bu4N[Ni(LSeO)2] (1) and Bu4N[Ni(LSO)2] (2) (H2LSeO = 3,5-di-tert-butyl-2-hydroxyselenophenol and H2LSO = 3,5-di-tert-butyl-2-hydroxythiophenol) were synthesised by reductive cleavage of the respective 2,2′-dichalcogenobis(4,6-di-tert-butylphenol) (H2LX–X; X = Se, S) with nickel(II) salts. The crystal structures of 1 and 2 confirm the reductive X–X bond cleavage with the concomitant formation of the corresponding monoanionic square planar complex, where quinoidal distortions of the aromatic rings are observed. The monoanionic complexes (1 and 2) are paramagnetic (S = 1/2), exhibiting rhombic EPR signals, and the g anisotropies are well correlated with the spin–orbit coupling of chalcogenides. The spectral data indicate that the ligands H2LXO in 1 and 2 are redox non-innocent and stabilise the square planar S = 1/2 nickel complexes with a highly delocalised unpaired electron. DFT calculations further support the delocalised electronic structures of the nickel complexes.


 Please wait while we load your content...
Please wait while we load your content...