Influence of organic cation planarity on structural templating in hybrid metal-halides†
Abstract
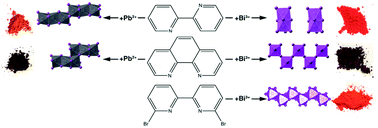
Controlling the connectivity and topology of solids is a versatile way to target desired physical properties. This is especially relevant in the realm of hybrid halide semiconductors, where the long-range connectivity of the inorganic substructural unit can lead to significant changes in optoelectronic properties such as photoluminescence, charge transport, and absorption. We present a new series of hybrid metal-halide semiconductors, (phenH2)BiI5·H2O, (2,2-bpyH2)BiI5, (BrbpyH)BiI4·H2O, (phenH2)2Pb3I10·2H2O, and (2,2-bpyH2)2Pb3I10 where (phenH2)2+ = 1,10-phenanthroline-1,10-diium, (2,2-bpyH2)2+ = 2,2′-bipyridine-1,1′-diium and (BrbpyH)+ = 6,6′-dibromo-2,2′-bipyridium. These compounds allow us to observe how the planarity of the cation, induced either through structural modification in the case of (phenH2)2+ or through non-covalent interactions in (BrbpyH)+, both relative to (2,2-bpyH2)2+, modifies the inorganic substructural unit. While the Pb2+ series of compounds show minimal changes in inorganic connectivity, we observe large differences in the Bi3+ series, ranging from 0-D dimers to corner- and edge-sharing 1-D chains of octahedra. We find that compounds containing (phenH2)2+ and (BrbpyH)+ pack more efficiently than those with (2,2-bpyH2)2+ due to their retention of planarity leading to greater inorganic connectivity. Electronic structure calculations and optical diffuse reflectance reveal that the band gaps of these compounds are influenced by the degree of inorganic connectivity and the inorganic substructural unit distances. These results show that the structure and planarity of organic cations can directly influence both the inorganic connectivity and the optical properties that could be tuned for certain optoelectronic applications.


 Please wait while we load your content...
Please wait while we load your content...