Heteroleptic ruthenium bis-terpyridine complexes bearing a 4-(dimethylamino)phenyl donor and free coordination sites for hydrogen photo-evolution†
Abstract
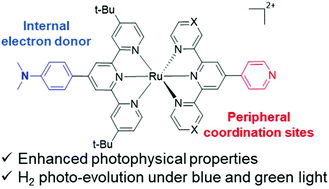
Motivated by the recent report of a heteroleptic ruthenium bis-terpyridine complex [Ru(toltpy)(bipytpy)](PF6)2 (toltpy: 4′-(tolyl)-2,2′:6′,2′′-terpyridine; bipytpy: 4′-(4-bromophenyl)-4,4′′′:4′′,4′′′′-di-pyridinyl-2,2′:6′,2′′-terpyridine) capable of driving the photo-evolution of hydrogen with a constant rate of activity for 12 days [M. Rupp et al., Inorg. Chem., 2019, 58, 9127–9134], we investigated the impact of an internal electron donor on the photoactivity of three new ruthenium bis-terpyridine photosensitizers. We used 4′-(N,N-dimethylaminophenyl)-4,4′′-di-tert-butyl-2,2′:6′,2′′-terpyridine (Dtpy) as the donor ligand. These complexes also bear peripheral coordination sites at various positions on the terpyridine backbone, allowing for photosensitizer–catalyst interactions. Their performances in photocatalysis under blue and green light (450 and 525 nm, respectively) were measured, in the presence of triethanolamine, using a cobaloxime catalyst [Co(dmgH)2(H2O)2](BF4)2. Complexes C1[Ru(Dtpy)(pytpy)](PF6)2 (pytpy: 4′-(pyridin-4-yl)-2,2′:6′,2′′-terpyridine) and C3[Ru(Dtpy)(bipytpy)](PF6)2 appeared to have a photostability similar to that of [Ru(toltpy)(bipytpy)](PF6)2 but with lower activity rates, while C2[Ru(Dtpy)(pz2bpy)](PF6)2 (pz2bpy: 2,6-di(pyrazin-2-yl)-4,4′-bipyridine) showed a better peak activity, however, followed by a progressive decay. After 24 hours, complexes C1, C2 and C3 had reached TONs of 18, 35 and 52 under blue light and 14, 20 and 47 under green light, respectively, and were still found to be active. Their photophysical and electronic properties are discussed to rationalize the photocatalytic trends.


 Please wait while we load your content...
Please wait while we load your content...