Direct comparison of dithienosilole and dithienogermole as π-conjugated linkers in photosensitizers for dye-sensitized solar cells†
Abstract
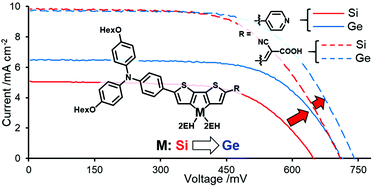
Dithienosilole (DTS) and dithienogermole (DTG) are useful building units of π-conjugated organic materials. In the present work, donor–π-acceptor (D–π-A) dyes with bis(dihexyloxyphenyl)aminophenyl, DTS or DTG, and pyridine or cyanoacrylic acid as the donor (D), the π-conjugated linker (π), and the acceptor (A) units, respectively, were prepared and their optical properties were investigated. The D–π-A dyes exhibited strong absorption in the visible region, indicating efficient intramolecular donor–acceptor interaction. The addition of trifluoroacetic acid to solutions of pyridine-containing dyes led to red-shifts of the absorption bands as a result of pyridinium salt formation. Similar red-shifts were observed for cyanoacrylic acid dyes, which were due to the enhanced formation of neutral dyes relative to the separated ion pairs. The D–π-A dyes, however, showed similar absorption spectra when attached to the TiO2 surface, indicating that the dye–TiO2 electronic interaction was rather weak. In contrast to the finding that these dyes exhibited similar optical properties regardless of the π-linker (i.e., DTS or DTG), dye-sensitized solar cells (DSSCs) based on DTG-containing dyes exhibited superior performance compared to those based on DTS-containing dyes. Electrochemical impedance spectroscopy measurements supported the higher performance of the DSSCs with DTG-containing dyes.


 Please wait while we load your content...
Please wait while we load your content...