Structural and spectroscopic analysis of a new family of monomeric diphosphinoboranes†
Abstract
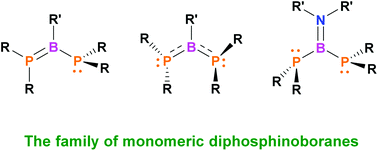
We present a series of amino- and aryl(diphosphino)boranes R2PB(R′′)PR′2, where R2P, R′2P = tBu2P, tBuPhP, Ph2P, Cy2P, and R′′ = iPr2N, Ph, which were obtained via the metathesis reaction of iPr2NBBr2 or PhBBr2 with selected lithium phosphides. The structures of isolated diphosphinoboranes were characterized in the solid state and in solution by means of X-ray diffraction and NMR spectroscopy, respectively. The utility of these P–B–P species as ligands for transition metal complexes was tested in the reaction with [(COD)PtMe2]. Moreover, we carried out DFT calculations to elucidate bonding interactions and philicity of the reactive centers as well as to analyze conformations of the studied species. Electronic and steric properties of substituents on P and B atoms were found to have a strong influence on the structures of the obtained compounds. Three main types of diphosphinoboranes were distinguished, based on the strength of P–B π-interaction within the molecule: (i) application of strong electron-donating substituents on P-atoms and electron-accepting phenyl groups on B atoms led to the structure with one double P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) B and one single P–B bond and diverse planar and pyramidal geometry of phosphanyl groups; (ii) reduction of the donor ability of phosphanyl groups gave diphosphanylboranes with delocalized P–B–P π-interactions; (iii) introduction of amino groups with strong donor abilities on B atoms canceled P–B π-interactions and allowed compounds with two very long P–B bonds and two pyramidal phosphanyl groups to be obtained.
B and one single P–B bond and diverse planar and pyramidal geometry of phosphanyl groups; (ii) reduction of the donor ability of phosphanyl groups gave diphosphanylboranes with delocalized P–B–P π-interactions; (iii) introduction of amino groups with strong donor abilities on B atoms canceled P–B π-interactions and allowed compounds with two very long P–B bonds and two pyramidal phosphanyl groups to be obtained.


 Please wait while we load your content...
Please wait while we load your content...