Tracking the dimensional conversion process of semiconducting lead bromide perovskites by mass spectroscopy, powder X-ray diffraction, microcalorimetry and crystallography†
Abstract
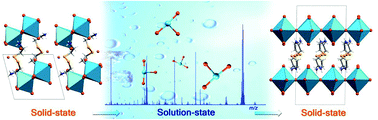
The structural information of a material in both the solid state and solution state is essential to the in-depth understanding of the properties of inorganic–organic hybrid materials. A one-dimensional (1D) lead bromide formulated as [H][NH3(CH2)2SS(CH2)2NH3][H2O][PbBr5] (1) could be converted into a new two-dimensional (2D) complex, [NH3(CH2)2SS(CH2)2NH3][PbBr4] (2), by soaking the crystals in water. The isolated 2D compound showed single-layer lead-halide perovskite structures. Electrospray ionization mass spectrometry (ESI-MS) analyses of the reaction solution revealed that the [PbBr3]− fragments are initially formed from the rapid decomposition of the 1D [PbBr5]3− chains and subsequently reassemble into 2D [PbBr4]2− layers, which was verified by powder X-ray diffraction (PXRD) and microcalorimetry. Because of the decomposition and reassembly process, complex 1 could be used as a precursor to synthesize M2+-doped 2D lead bromide perovskites, namely, Mn@2, Ni@2 and Cd@2. In addition, preliminary tests indicated that complex 2 exhibited a lower optical band gap (3.25 eV) and higher electrical conductivity (3.2 × 10−11 S cm−1) than complex 1 (3.38 eV, 5.4 × 10−12 S cm−1).


 Please wait while we load your content...
Please wait while we load your content...