31P NMR study of the activated radioprotection mechanism of octylphenyl-N,N-diisobutylcarbamoylmethyl phosphine oxide (CMPO) and analogues†
Abstract
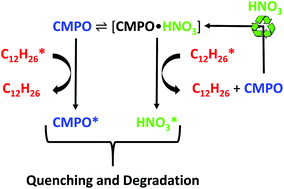
We report a 31P NMR investigation into the activated radioprotection mechanism of octylphenyl-N,N-diisobutyl-carbamoylmethyl phosphine oxide (CMPO) and analogues in the presence of a gamma radiation field. CMPO exhibits significantly enhanced radiation resistance in the presence of high nitric acid concentrations, compared to other ligands proposed for recovery of the trivalent actinides from spent nuclear fuel. The fundamental mechanism behind this activated radioprotection has been investigated using 31P NMR and other supporting analytical techniques (GCMS and LCMS) in conjunction with systematic gamma irradiation studies, covering solvent system formulation and structural effects through the use of the CMPO analogues, dioctylphenylphospine oxide (DOPPO) and trioctylphosphine oxide (TOPO). These experiments have demonstrated that the acid-dependent, radioprotection mechanism requires a protonated phenyl-phosphine oxide motif to activate. Further, contacting these three ligand loaded organic phases with a range of mineral acids (nitric, sulfuric, hydrochloric, and perchloric acids) shows specificity for nitric acid (HNO3), and the formation of a distinct [ligand·HNO3] complex for CMPO and DOPPO, as identified by 31P NMR, and predicted by DFT calculations. We propose that this complex is capable of sequential n-dodecane excited state quenching through the conjugated aromatic functionalities on the constituent CMPO and DOPPO molecules.


 Please wait while we load your content...
Please wait while we load your content...