Dinuclear helicate and tetranuclear cage assembly using appropriately designed ditopic triazole-azine ligands†‡
Abstract
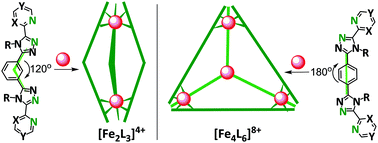
Four new, symmetrical, bis-bidentate ditopic Rdpt-type, Rat (R azine-triazole), ligands have been prepared, Lnpym-meta/para (n = 2 or 4), which contain bidentate n-pyrimidine/triazole binding pockets connected through an appropriate aromatic spacer, meta/para-phenyl, to enable assembly into dinuclear helicates or tetranuclear tetrahedral cages, respectively. The 3 : 2 self assembly reactions of each Lnpym-meta/para ligand with iron(II) tetrafluoroborate gave the desired complexes, as shown by X-ray crystal structure determinations of the pair of helicates [FeII2(Lnpym-meta)3(BF4)4]·6CH3CN, with n = 2 (1·6CH3CN) or 4 (2·6CH3CN), and the pair of Td cages [FeII4(Lnpym-para)6(BF4)8]·xsolvent, with n = 2 (3·xsolvent) or 4 (4·xsolvent). Reversible FeII/III processes at Em = 0.95 ± 0.05 V vs. 0.01 M AgNO3/Ag in MeCN are a feature of 1–4, with little variation in the redox potential as a function of nuclearity, architecture or choice of n-pyrimidine isomer. In all four complexes the iron(II) centres are low spin, despite having employed the weakest field diazines, the 2- and 4-pyrimidines, in these ditopic Rat ligands. Nevertheless this is an exciting proof of principle that Rdpt-type ligands (previously used to generate about 50 spin crossover-active complexes) can be extended into ditopic forms that are suitable for supramolecular assembly of helicates and cages.
- This article is part of the themed collection: Modern coordination chemistry


 Please wait while we load your content...
Please wait while we load your content...