The study of Rh(iii) hydroxocomplexes using capillary zone electrophoresis with a UV–Vis detector: the development of the method†
Abstract
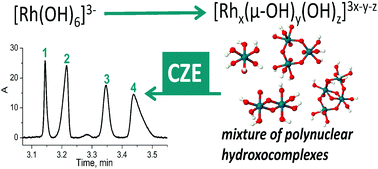
The expediency of capillary zone electrophoresis application in the study of a mixture of rhodium(III) hydroxocomplexes is shown for the species formed during the thermal treatment of [Rh(OH)6]3− at 60 °C in alkaline media. As these compounds are unstable in solution because of polycondensation at pH > 10 and the formation of insoluble rhodium(III) hydroxides at pH 5–10, their acidic derivatives with terminal aqua ligands acted as the objects of the study. Optimal separation conditions were achieved using sodium perchlorate as a background electrolyte, a voltage of +20 kV and pH 2.6. Rhodium(III) species with different nuclearities were identified in accordance with their UV spectra and the regularities of electrophoretic migration in the capillary. The presence of a dimer was also verified by the spiking of the mixture under investigation with a previously synthesised complex [Rh2(μ-OH)2(H2O)8](NO3)4, which confirms the correct identification.


 Please wait while we load your content...
Please wait while we load your content...