A light activated CMP conjugated 8-aminoquinoline turn-on fluorescent optode for selective determination of Th4+ in an aqueous environment†
Abstract
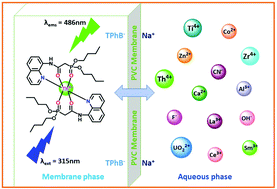
A new dibutyl(2-oxo-2-(quinolin-8-ylamino)ethyl)phosphinate (L) was designed, synthesised and developed as a light activated optode for Th4+ determination. The sensing performance of L was studied in solution and in the polymeric membrane phase using absorbance and emission techniques. The neutral ion carrier L exhibits weak fluorescence at 400 nm in CH3CN : H2O (9 : 1, v/v). Upon complexation of L with Th4+, the emission intensity at 486 nm was increased 100-fold due to chelation induced enhanced fluorescence. The L–Th4+ binding interaction was studied using both Hill's and Job's plots. They indicate the formation of complex species in a 2 : 1 ratio with an estimated binding constant of 2.66 × 104 M−2. Complex formation between L and Th4+ was studied by NMR (1H, 31P), FTIR and LC-MS analyses. In order to synthesize a probe for the sensing of Th4+ in 100% aqueous medium, the optode was prepared by immobilizing L as a neutral ion carrier in a PVC support using dioctyl phthalate (DOP) as a membrane solvent. The best performance of the optode was observed with a membrane composition of PVC : L : NaTPB : DOP in proportions of 35 : 5 : 3 : 57 (%, w/w) in the pH range of 4.0–8.0. The optode can detect Th4+ concentrations down to 1.1 nM with a fast response time of 15 s and the optical response remains unaltered even after 3 months; the optode sensor can be regenerated using Na2EDTA solution. Finally, the optode was used for the quantification of Th4+ in various water samples, monazite sand, and gas mantle samples.


 Please wait while we load your content...
Please wait while we load your content...