Sensing alcohol vapours with novel unsymmetrically substituted metallophthalocyanines
Abstract
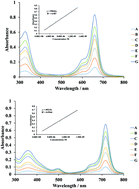
Unsymmetrically substituted phthalocyanines were synthesized by the well-known statistical condensation method using two differently substituted precursors, 4-tert-butylphthalonitrile and 4-(4-pyrrol-1-yl)phenoxyphthalonitrile. Spin-coated thin films of these compounds were subsequently exposed to varying concentrations of methanol, ethanol and butanol between 25 and 150 ppm in order to investigate the effects of the number of carbon atoms in the alcohol vapors. The results of this preliminary investigation indicated that sensor performance parameters such as sensitivity, response and recovery time strongly depend on the number of carbon atoms in the analyte molecules. It was found that the sensitivity of the sensors decreases with increasing number of carbon atoms. Electrochemistry studies of CoPc and MnClPc were carried out with cyclic and square wave voltammetry methods. Both complexes illustrated metal and ring-based electron transfer reactions. While CoPc underwent only one metal-based reduction ([CoIIPc2−]/[CoIPc2−]1−), two metal-based reductions ([Cl–MnIIIPc2−]/[Cl–MnIIPc2−]1− and [Cl–MnIIPc2−]1−/[Cl–MnIPc2−]2−) were observed with MnPc. Color changes during the in situ spectroelectrochemical measurements illustrated the versatility of the compounds for display technologies. Pyrrole-containing substituents of the complexes triggered their electropolymerization on the working electrode, which enhances their value as functional materials for modified electrodes.


 Please wait while we load your content...
Please wait while we load your content...