A new approach to spectroscopic and structural studies of the nano-sized silicate-substituted hydroxyapatite doped with Eu3+ ions
Abstract
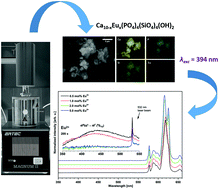
Nanocrystalline silicate-substituted hydroxyapatites Ca10−xEux(PO4)4(SiO4)2(OH)2 (where x = 0.5, 1.0, 2.0, 5.0 mol%) doped with Eu3+ ions were synthesized using a microwave assisted hydrothermal method and heat-treated in the temperature range from 700 to 1000 °C. The concentration of optically active Eu3+ ions was established in the range of 0.5–5 mol% to investigate the preference of occupancy sites. The structural and morphological properties of the obtained biomaterials were determined by using XRD (X-Ray Powder Diffraction), TEM (Transmission Electron Microscopy) and SEM (Scanning Electron Microscopy) techniques as well as infrared (IR) spectroscopy. The average particle sizes were calculated to be in the range from 20 nm to 80 nm by the Rietveld method. The charge compensation mechanism in europium(III)-doped silicate-substituted hydroxyapatite was proposed in the Kröger–Vink-notation. The luminescence properties (the emission, excitation spectra and emission kinetics) of the Eu3+ ion-doped apatite were recorded depending on the dopant concentration. The existence of Eu2+ ions was confirmed by the emission spectra.


 Please wait while we load your content...
Please wait while we load your content...