A tris-(manganese(iii))corrole–porphyrin–corrole triad: synthesis, characterization and catalytic epoxidation†
Abstract
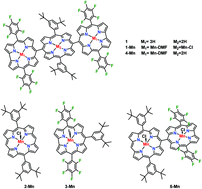
A homotrimetallic manganese(III) corrole–porphyrin–corrole triad has been synthesized and structurally characterized. Corrole and porphyrin rings in the free base corrole–porphyrin–corrole triad are found to be roughly perpendicular to each other having a dihedral angle of 83° in the solid state. A dihedral angle of nearly 67° was found between Mn(III)-corroles and Mn(III)-porphyrin rings in the manganese triad complex, 1-Mn. The absorption spectrum of the manganese triad was observed to be the sum of the absorptions from manganese corrole and manganese porphyrin. The catalytic activity of the manganese triad complex was investigated for the epoxidation of styrene using iodobenzene diacetate as an oxygen source. Preliminary results obtained showed the superior activity of the manganese triad as an oxidation catalyst as compared to the respective monomer catalysts. Moreover, product yield in the oxidation of styrene catalyzed by the manganese triad was found to be higher than that of the combined activity of both monomers taken in same equivalence. The reason behind this higher yield may be attributed to an increased local concentration of the catalytic centers at the reaction site.
- This article is part of the themed collection: New Talent: Asia-Pacific


 Please wait while we load your content...
Please wait while we load your content...