Isolation of ligand-centered borocations in molybdenum complexes containing a triaminoborane-bridged diphosphorus ligand†
Abstract
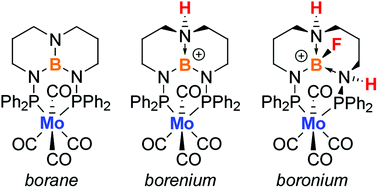
Metal complexes that form isolable, ligand-centered borenium ions (i.e. reactive three-coordinate boron cations) are rare, especially with highly-versatile diphosphorus ligands. Here we report the first structurally-characterized examples of ligand-centered borocations in a class of diphosphorus ligands derived from the bicyclic triaminoborane 1,8,10,9-triazaboradecalin (TBD). Treating (PhTBDPhos)Mo(CO)4 (1) with HOTf or HNTf2 resulted in protonation of the bridgehead α-nitrogen on the TBD backbone and formation of ligand-centered borenium ions (1-HOTf and 1-HNTf2, respectively), whereas reaction of 1 with HBF4·Et2O resulted in protonation of two α-nitrogen atoms and fluoride abstraction from BF4− to form a four-coordinate borocation in [1-H2F][BF4]. Single-crystal XRD data confirmed the formation of the borocations, and multinuclear NMR and IR spectroscopy studies were used to interrogate the electronic environment at molybdenum and boron. The 11B NMR resonances for the borenium ions in 1-HOTf and 1-HNTf2 (δ 29.5 and 29.6 ppm) were more deshielded than the resonance for 1 (δ 25.9 ppm), consistent with the decreased electron density at boron. The 31P NMR data revealed similar trends in response to increasing protonation on the TBD backbone, and aligned well with small, stepwise increases in Mo–CO stretching frequencies that followed the order borane (1) < borenium (1-HOTf and 1-HNTf2) < boronium ([1-H2F][BF4]). Density functional theory calculations conducted on 1, 1-HOTf, and [1-H2F][BF4] revealed subtle changes in boron and nitrogen atomic charges consistent with those calculated for more well-established borenium ions in metal-free systems. Overall, the results confirm previous observations of latent borenium ion reactivity in TBDPhos complexes.
- This article is part of the themed collection: Inorganic chemistry of the p-block elements


 Please wait while we load your content...
Please wait while we load your content...