Evaluation of attractive interactions in the second coordination sphere of iron complexes containing pendant amines†
Abstract
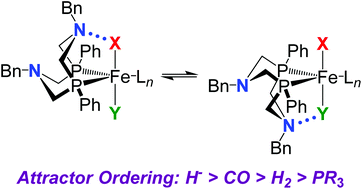
The interactions between pendant amines in the second coordination sphere and ligands in the first coordination sphere are important for understanding the structures and reactivity of complexes containing PR2NR′2 ligands, which have been shown to be highly active H2 oxidation/production catalysts. A series of [Fe(PPh2NBn2)2(X)(Y)]n+ complexes have been prepared and structurally characterized. These complexes have two different ligands with which the pendant amines of the diphosphine ligand can interact. The solid state structure of cis-Fe(PPh2NBn2)2Cl2 reveals that the six-membered rings adjacent to the P atoms are in a boat confirmation, resulting in close N⋯P distances that suggests the P atoms have a greater affinity for the lone pair of electrons on the N atom than chloride ligands. Similarly, boat conformations are observed for both rings adjacent to the hydride ligands of trans-[HFe(PPh2NBn2)2(CH3CN)]+ and trans-HFe(PPh2NBn2)2Cl, resulting in short N⋯H distances. Spectroscopic and computational studies of trans-[HFe(PPh2NBn2)2(CO)]+, trans-[HFe(PPh2NBn2)(PPh2NBn2H)(CO)]2+, and trans-[HFe(PPh2NBn2)2(H2)]+ indicate the complexes are more stable when the pendant amines in boat conformations are adjacent to the hydride ligand. These data suggest an attractor ordering of H− > CO > H2 > PR3 > Cl− ∼ CH3CN.
- This article is part of the themed collection: Inorganic chemistry of the p-block elements


 Please wait while we load your content...
Please wait while we load your content...