Coordination design of cadmium ions at the 4-fold axis channel of the apo-ferritin cage†
Abstract
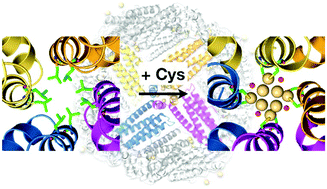
Spherical protein cages with highly symmetrical structures provide unique environments for the conjugation of metal ions and metal nanoparticles. Ferritin has been widely studied as a template for the coordination of metal ions and metal nanoparticles in fundamental research and applications. However, it remains difficult to design metal coordination sites precisely. In this work, we describe the design and construction of new metal coordination sites by introducing Cys residues at the 4-fold symmetrical hydrophobic channel of apo-ferritin. X-ray crystal structure analyses of the mutants containing Cd(II) ions show that the four or eight binding sites for Cd(II) ions are located at the 4-fold symmetrical axis channel of apo-ferritin. It was found that the coordination number and configuration of Cd(II) ions can be varied by adjusting the positions of the Cys residues at the symmetrical channels of the apo-ferritin cage.
- This article is part of the themed collection: The central role of the d-block metals in the periodic table


 Please wait while we load your content...
Please wait while we load your content...