Molybdenum(ii) complexes with p-substituted BIAN ligands: synthesis, characterization, biological activity and computational study†
Abstract
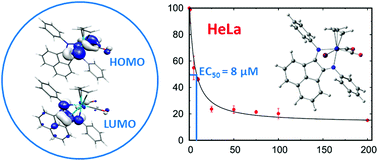
New complexes [Mo(η3-C3H5)X(CO)2(4-Y-BIAN)] (4-Y-BIAN = bis(4-Y-phenyl)-acenaphthenequinonediimine), with X = Br and Y = H, Me, OMe, COOH and X = Cl, Y = OMe, as well as the cation with X = NCMe and Y = OMe were synthesized, expanding the scope of this family. Two single crystal X-ray structures (X = Br, Y = Me, OMe) display a less symmetric arrangement (axial isomer), where one N donor atom is trans to the allyl group and the second to one CO. DFT studies showed similar energies for the two possible isomers of the complexes, with a very small preference for the observed axial isomer. The HOMO of the complexes is localized in the metal and the HOMO−1 of the oxidized species has a contribution from the BIAN ligand, while the LUMO is fully localized in BIAN. Electrochemical studies showed one process corresponding to the oxidation of Mo(II) to Mo(III) for complexes with X = Br, Y = H, Me, and two oxidation reactions for those with X = Br, Y = Cl, OMe, while the COOH derivative exhibited no oxidation wave. The antitumor effect of the complexes with X = Br was tested in cancer lines, and the H and OMe complexes were particularly active, with EC50 values below 8 μM in HeLa cell lines. The DNA binding constants determined by titration experiments were comparable with those of doxorubicin and ethidium bromide, suggesting a mechanism of action based on intercalation in DNA.
- This article is part of the themed collection: Nitrogen Ligands


 Please wait while we load your content...
Please wait while we load your content...