Increasing steric demand through flexible bulk – primary phosphanes with 2,6-bis(benzhydryl)phenyl backbones†
Abstract
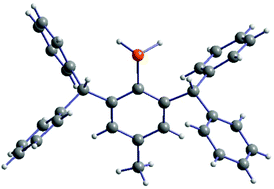
Sterically demanding primary phosphanes of the type RBhp-PH2 (Bhp = 2,6-bis(benzhydryl)-4-R-phenyl; R = Me, tBu) could be prepared in high yields by modification of synthetic protocols of established bulky phosphanes. The Bhp substituents simultaneously exhibit extensive steric expansiveness and high degrees of flexibility compared to other 2,6-substituted phenyl backbones, enabling a range of different-sized atoms and functionalities to be located between the flanking benzhydryl moieties. Therefore, it was also possible to isolate and fully characterize several halogenated precursors, a diazonium intermediate, as well as a dihalostibane.
- This article is part of the themed collection: Inorganic chemistry of the p-block elements


 Please wait while we load your content...
Please wait while we load your content...