The influence of metal-complexing macrocycle size on intramolecular movement in rotaxanes†
Abstract
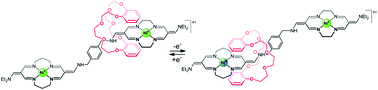
[2]rotaxanes composed of a dibenzo-24-crown-8 wheel (DB24C8) and an axle containing two metal-complexing tetraazamacrocyclic units linked with a p-xylylene bridge, coordinating the same or different metal ions (Ni and/or Cu) are described. A symmetric di-Ni rotaxane crystallizes in the monoclinic P21/c space group with one rotaxane cation and four PF6− counterions in the asymmetric part of the unit cell. In the crystal, the cations and anions of the investigated compound form an intertwined 3D-framework with C–H⋯F and C–H⋯π intermolecular interactions. The NMR ROESY spectrum of the diamagnetic di-Ni rotaxane confirms that at room temperature slow shuttling of the DB24C8 moiety takes place along the symmetric axle. The non-symmetric rotaxane, containing 14- and 16-membered nickel(II)-complexing macrocycles, exists in solution as an equilibrium mixture of two conformers in the ratio of ca. 1 : 7. This rotaxane behaves as a potential-controlled molecular switch: in the major isomer, the DB24C8 moiety is localized in the vicinity of the 14-membered metal-complexing macrocycle, and moves towards the 16-membered unit upon the first oxidation. Reverse translocation occurs after the subsequent oxidation of the 14-membered complex unit. In contrast, no such movements of the DB24C8 moiety were observed upon investigation of the heteronuclear [2]rotaxane, coordinating copper(II) and nickel(II) ions.


 Please wait while we load your content...
Please wait while we load your content...