FLP behaviour of cationic titanium complexes with tridentate Cp,O,N-ligands: highly efficient syntheses and activation reactions of C–X bonds (X = Cl, F)†
Abstract
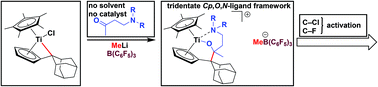
The synthesis of cationic titanium complexes 4a,b with tridentate Cp,O,N-ligand frameworks, starting from the monopentafulvene complex Cp*Ti(Cl)(π-η5:σ-η1-C5H4CR2 (CR2 = adamantylidene) (1) and bidentate O,N-ligand precursors CH3C(O)CH2CH2NR2 (R = Et, CH2Ph) (L1a,b), in a high-yielding and efficient two-step synthetic pathway is described. The β-aminoketones L1a,b were synthesized by a herein reported solvent- and catalyst-free reaction. The reaction pathway involves insertion reactions, subsequent methylations and final activations with B(C6F5)3. NMR investigations of the cationic titanium complex 4a in deuterated dichloromethane revealed an ongoing selective reaction under formation of the cationic complex 5a-d2, which is the result of C–Cl bond cleavage. In addition to selective C–Cl bond activation reactions, C(sp3)–F bonds were activated by 4a,b, pointing out the special tm-FLP nature of 4a,b.


 Please wait while we load your content...
Please wait while we load your content...