Observation of visible light activated photocatalytic degradation of stearic acid on thin films of tantalum oxynitride synthesized by aerosol assisted chemical vapour deposition†‡
Abstract
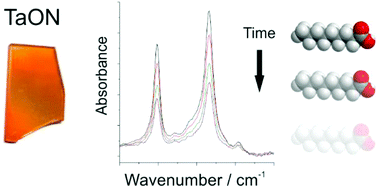
UV activated photocatalysts deposited using chemical vapour deposition have found commercial success as self-cleaning coatings. However, only limited work has been conducted on the use of the more recently discovered visible light activated photocatalysis for this application. Tantalum oxynitride is an established visible light photocatalyst, and in this paper we have investigated the ability of thin films of tantalum oxynitride to photocatalytically degrade a model organic pollutant, stearic acid, and therefore assess the coatings potential for self-cleaning applications. Thin films of tantalum oxide were formed using aerosol assisted chemical vapour deposition (AACVD) of tantalum ethoxide, and then converted into tantalum oxynitride through ammonolysis at temperatures between 550 °C and 750 °C. Investigation of the films using XRD, UV-vis spectroscopy and XAFS identify that amorphous tantalum oxynitride is formed during the ammonolysis, with complete conversion to TaON under conditions of 700 °C for 24 hours. The self-cleaning ability of this film was assessed using stearic acid as the model pollutant, with a degradation rate of 2.5(2) × 1013 molecules per min per cm2 when exposed to a 5-sun solar simulator, equipped with a UV cut-off filter. We therefore conclude that tantalum oxynitride thin films are able to act as self-cleaning coatings through visible light photocatalysis and that films of tantalum oxynitride can be synthesized using a scalable chemical vapour deposition route.


 Please wait while we load your content...
Please wait while we load your content...