Dioxygen activation by a dinuclear thiolate-ligated Fe(ii) complex†
Abstract
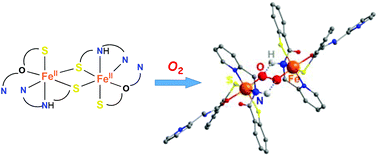
Dioxygen activation by FeII thiolate complexes is relatively rare in biological and chemical systems because the sulfur site is at least as vulnerable as the iron site to oxidative modification. O2 activation by FeII–SR complexes with thiolate bound trans to the O2 binding site generally affords the FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O intermediate and oxidized thiolate. On the other hand, O2 activation by Fe(II)–SR complexes with thiolate bound cis to the O2 binding site generates FeIII–O–FeIII or S-oxygenated complexes. The postulated FeIV
O intermediate and oxidized thiolate. On the other hand, O2 activation by Fe(II)–SR complexes with thiolate bound cis to the O2 binding site generates FeIII–O–FeIII or S-oxygenated complexes. The postulated FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O intermediate has only been identified in isopenicillin N synthase recently. We demonstrated here that O2 activation by a dinuclear FeII thiolate-rich complex produces a mononuclear FeIII complex and water with a supply of electron donors. The thiolate is bound cis to the postulated dioxygen binding site, and no FeIII–O–FeIII or S-oxygenated complex was observed. Although we have not detected the transient intermediate by spectroscopic measurements, the FeIV
O intermediate has only been identified in isopenicillin N synthase recently. We demonstrated here that O2 activation by a dinuclear FeII thiolate-rich complex produces a mononuclear FeIII complex and water with a supply of electron donors. The thiolate is bound cis to the postulated dioxygen binding site, and no FeIII–O–FeIII or S-oxygenated complex was observed. Although we have not detected the transient intermediate by spectroscopic measurements, the FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O intermediate is suggested to exist by theoretical calculation, and P-oxidation and hydride-transfer experiments. In addition, an unprecedented FeIII–O2–FeIII complex supported by thiolates was observed during the reaction by using a coldspray ionization time-of-flight mass (CSI-TOF MS) instrument. This is also supported by low-temperature UV-vis measurements. The intramolecular NH⋯O
O intermediate is suggested to exist by theoretical calculation, and P-oxidation and hydride-transfer experiments. In addition, an unprecedented FeIII–O2–FeIII complex supported by thiolates was observed during the reaction by using a coldspray ionization time-of-flight mass (CSI-TOF MS) instrument. This is also supported by low-temperature UV-vis measurements. The intramolecular NH⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) FeIV hydrogen bonding, calculated by DFT, probably fine tunes the O2-activation process for intramolecular hydrogen abstraction, avoiding the S-oxygenation at cis-thiolate.
FeIV hydrogen bonding, calculated by DFT, probably fine tunes the O2-activation process for intramolecular hydrogen abstraction, avoiding the S-oxygenation at cis-thiolate.


 Please wait while we load your content...
Please wait while we load your content...