2,6-Bis(benzimidazol-2-yl)pyridines as more electron-rich and sterically accessible alternatives to 2,6-bis(imino)pyridine for group 13 coordination chemistry†
Abstract
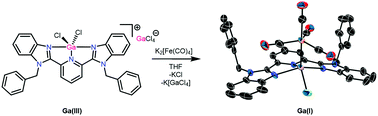
Treatment of GaCl3 with 2,6-bis(benzimidazol-2-yl)pyridine (G-BZIMPY, G = NBz, N(3,5-CF3)Bz, N-allyl and O) yielded the autoionization products [G-BZIMPYGaCl2][GaCl4] (1–4) in great yields. The Ga(III) complex 1 was reduced to Ga(I) using K2[Fe(CO)4], resulting in the complex [(NBzBZIMPY)(Cl)Ga–Fe(CO)4] (7). GaCl3 and AlCl3 were complexed by the structurally similar bis(imino)pyridine (DIMPY) and the resulting complexes are compared to those of G-BZIMPY.


 Please wait while we load your content...
Please wait while we load your content...