Design considerations for chiral frustrated Lewis pairs: B/N FLPs derived from 3,5-bicyclic aryl piperidines†
Abstract
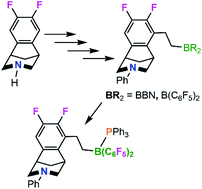
Herein, 3,5-bicyclic aryl piperidines are derivatized to generate chiral B/N FLPs. Initially, the twofold symmetric amine C6H2F2(C5H8NiPr) 1 was converted in a series of synthetic steps to the styrene-derivative C6HF2(C5H8NiPr)(CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2) 4. Efforts to hydroborate the vinyl fragment proved challenging as a result of the strongly basic nitrogen, although the species C6HF2(C5H8N(H)iPr)(CH2CH2B(OH)(C6F5)2) 5 was crystallographically characterized. Modification of the system was achieved by conversion of the amine C6H2F2(C5H8NH) 6 to C6HF2(C5H8NPh)(CH
CH2) 4. Efforts to hydroborate the vinyl fragment proved challenging as a result of the strongly basic nitrogen, although the species C6HF2(C5H8N(H)iPr)(CH2CH2B(OH)(C6F5)2) 5 was crystallographically characterized. Modification of the system was achieved by conversion of the amine C6H2F2(C5H8NH) 6 to C6HF2(C5H8NPh)(CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2) 9. Hydroboration of 9 with 9-BBN or HB(C6F5)2 gave C6HF2(C5H8NPh)(CH2CH2BBN) 10 or C6HF2(C5H8NPh)(CH2CH2B(C6F5)2) 11, respectively. The latter species was derivatized by complexation of PPh3 to give C6HF2(C5H8NPh)(CH2CH2B(C6F5)2)(PPh3) 12. The Lewis acidities of 10 and 11 were assessed by the Gutman–Beckett test and by computations of the FIA and GEI. While 10 did not effect HD scrambling or hydrogenation of N-phenylbenzylimine, 11 was effective in HD scrambling. Despite this, no reduction of N-t-butylbenzylimine or N-phenylbenzylimine was achieved. These data demonstrate that 10 lacks the threshold combination of Lewis acidity and basicity to activate H2, while 11 lacks the steric demands about boron to preclude classical Lewis acid–base bond formation with imine substrates.
CH2) 9. Hydroboration of 9 with 9-BBN or HB(C6F5)2 gave C6HF2(C5H8NPh)(CH2CH2BBN) 10 or C6HF2(C5H8NPh)(CH2CH2B(C6F5)2) 11, respectively. The latter species was derivatized by complexation of PPh3 to give C6HF2(C5H8NPh)(CH2CH2B(C6F5)2)(PPh3) 12. The Lewis acidities of 10 and 11 were assessed by the Gutman–Beckett test and by computations of the FIA and GEI. While 10 did not effect HD scrambling or hydrogenation of N-phenylbenzylimine, 11 was effective in HD scrambling. Despite this, no reduction of N-t-butylbenzylimine or N-phenylbenzylimine was achieved. These data demonstrate that 10 lacks the threshold combination of Lewis acidity and basicity to activate H2, while 11 lacks the steric demands about boron to preclude classical Lewis acid–base bond formation with imine substrates.
- This article is part of the themed collection: Inorganic chemistry of the p-block elements


 Please wait while we load your content...
Please wait while we load your content...