Fluorescent functionalised naphthalimides and their Au(i)–NHC complexes for potential use in cellular bioimaging†‡
Abstract
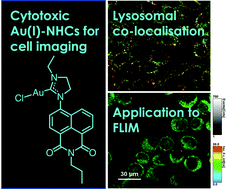
A series of cationic, dihydroimidazolinium-functionalized 1,8-naphthalimide fluorophores have been isolated as their hexafluorophosphate salts, [HL]PF6. These pro-ligands react with [AuCl(tht)] in the presence of base to form N-heterocyclic carbene (NHC) complexes, [AuCl(L)]. Two X-ray structures represent a pro-ligand and complex pairing: the latter reveals the two-coordinate linear geometry of the NHC–Au(I) species, as well as intermolecular interactions supported by both ligand π–π stacking and a weak aurophilic interaction of 3.3205(6) Å. The luminescence properties of the pro-ligands and complexes are dominated by the ICT character of the substituted fluorophore at ca. 500 nm, which is further modulated via functionalization at the 4-position of the naphthalimide. Cytotoxicity assessments were performed for all [HL]PF6 and [AuCl(L)] species against LOVO, MCF-7, A549 and PC3 cell lines; added lipophilicity seems to correlate with increased cytotoxicity. Confocal fluorescence microscopy was undertaken on a selected [HL]PF6 and [AuCl(L)] species and showed that the intracellular distribution is dependent upon the specific ligand structure. More detailed co-localisation studies show that selected examples present a predominant lysosomal staining pattern. FLIM studies exemplified the applicability of these probes, and secondly suggest that fluorescence lifetime could be used to provide information on the integrity of the complex and thus liberation of gold in a biological environment.


 Please wait while we load your content...
Please wait while we load your content...