Tetranuclear cobalt(ii)–isonicotinic acid frameworks: selective CO2 capture, magnetic properties, and derived “Co3O4” exhibiting high performance in lithium ion batteries†
Abstract
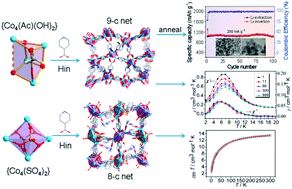
Two new 3D cobalt metal–organic frameworks (MOFs), [Co4(CH3COO)(in)5(μ3-OH)2]·2H2O (1) and [Co4(SO4)2(in)4(DMF)2]·3DMF (2) (Hin = isonicotinic acid), have been prepared through the anion template method. Compound 1 consists of rare odd-number connected (9-connected) cubane-like SBUs, while compound 2 consists of 8-connected high-symmetry square-planar clusters. Magnetic studies indicate that compound 1 exhibits spin-canting antiferromagnetic ordering, while compound 2 shows antiferromagnetic behavior. At 273 K and 1 bar, compound 1 exhibits a high CO2 selectivity over CH4 and a significant CO2 uptake of 13.6 wt%, which is higher than that of 2 (8.5 wt%). Furthermore, compound 1 was then transformed into ultrasmall Co3O4 nanoparticles via simple but effective annealing treatment. Electrochemical measurements show that the Co3O4 nanospheres derived from compound 1 exhibited high and stable lithium storage properties (1100 mA h g−1 after 100 cycles at 200 mA g−1) and excellent rate capabilities.


 Please wait while we load your content...
Please wait while we load your content...