Different phases of few-layer MoS2 and their silver/gold nanocomposites for efficient hydrogen evolution reaction†
Abstract
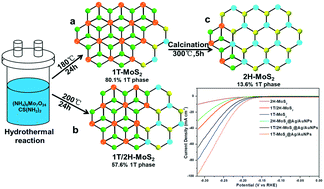
Molybdenum disulfide (MoS2) is expansively studied as the most promising non-precious metal catalyst for the hydrogen evolution reaction (HER) because of its excellent properties. However, the properties of MoS2 are quite distinct when the contents of the trigonal (1T) phase and hexagonal (2H) phase constituting MoS2 are different. Herein, three MoS2 representatives with different contents of the 1T phase are synthesized by a simple method, denoted as 1T-MoS2, 1T/2H-MoS2 and 2H-MoS2 from high to low contents of the 1T phase. The chemical composition and crystalline phase of the three samples are systematically analyzed and characterized. To improve charge transport between adjacent nanosheets of MoS2, silver and gold nanoparticles (Ag/AuNPs) are decorated on MoS2, denoted as 1T-MoS2@Ag/AuNPs, 1T/2H-MoS2@Ag/AuNPs and 2H-MoS2@Ag/AuNPs. The HER catalytic experiments of various MoS2 samples in 0.5 M H2SO4 show that 1T-MoS2 has the best catalytic performance compared to 1T/2H-MoS2 and 2H-MoS2. Owing to the cooperative effect of the 1T phase and Ag/AuNPs, the 1T-MoS2@Ag/AuNP nanocomposite exhibits better HER electrocatalytic activity than the other MoS2 samples because it contains a higher-percentage 1T phase and a higher charge transfer rate. This study offers an abundant foundation for designing and improvement of electrocatalysts based on MoS2 and its nanocomposites.


 Please wait while we load your content...
Please wait while we load your content...