Catalytic mechanisms of oxygen-containing groups over vanadium active sites in an Al-MCM-41 framework for production of 2,5-diformylfuran from 5-hydroxymethylfurfural†
Abstract
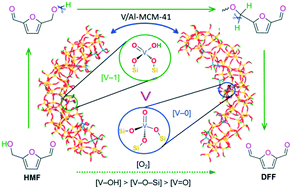
V-containing catalysts exhibit good catalytic performance toward the selective oxidation of 5-hydroxymethylfurfural (HMF) to 2,5-diformylfuran (DFF). Here, we report our study on the catalytic mechanism of –(SiO)3V(O) ([V-0]) and –(SiO)2V(O)(OH) ([V-1]) on a V-doped Al-MCM-41 pore model (V/Al-MCM-41) for the aerobic oxidation of HMF to DFF. For the two active sites, there are three types of oxygen-containing functional groups, which are hydroxyl-oxygen ([V–OH]), lattice-oxygen ([V–O–Si]), and terminal-oxygen ([V![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O]). We show that the catalytic cycle involves two HMF molecules, and there are mainly two activation steps, i.e., both O–H and C–H bond cleavages of HMF, and the rate-determining step is associated with the C–H bond cleavage of the first HMF molecule. We illustrate the efficiency of the catalytic contribution as [V–OH] > [V–O–Si] > [V
O]). We show that the catalytic cycle involves two HMF molecules, and there are mainly two activation steps, i.e., both O–H and C–H bond cleavages of HMF, and the rate-determining step is associated with the C–H bond cleavage of the first HMF molecule. We illustrate the efficiency of the catalytic contribution as [V–OH] > [V–O–Si] > [V![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O], and the [V-1] active site with a hydroxyl group displays higher catalytic activity than the [V-0] active site without a hydroxyl group. The present study not only brings an in-depth understanding of the activation of both O–H and C–H bonds which has been proposed based on experimental results for biomass molecules, but also makes one step forward toward the mechanism-guided design and synthesis of efficient, environmentally-friendly, and low temperature recyclable heterogeneous catalysts.
O], and the [V-1] active site with a hydroxyl group displays higher catalytic activity than the [V-0] active site without a hydroxyl group. The present study not only brings an in-depth understanding of the activation of both O–H and C–H bonds which has been proposed based on experimental results for biomass molecules, but also makes one step forward toward the mechanism-guided design and synthesis of efficient, environmentally-friendly, and low temperature recyclable heterogeneous catalysts.


 Please wait while we load your content...
Please wait while we load your content...