OH/Na co-functionalized carbon nitride: directional charge transfer and enhanced photocatalytic oxidation ability†
Abstract
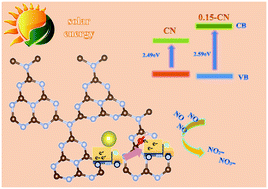
Graphitic carbon nitride (g-C3N4, CN for short) is a compelling visible-light responsive photocatalyst. However, its photocatalytic efficiency is low due to the random carrier transfer in planes and insufficient redox potential. Herein, we build oxygen functional group modified sodium-doped carbon nitride (OH/Na co-functionalized carbon nitride) to promote directional transfer of charge carriers for acceleration of separation and enhance redox potential for efficient oxidation of NO in air. Specifically, the function of sodium atoms could control the directional transfer of random carriers from the intralayer to the oxygen functional group-modified surface for the purpose of effectively reducing photogenerated electron–hole recombination. Meanwhile, the modification by oxygen-containing functional groups could adjust the band structure of CN, thereby increasing the oxidation–reduction potential of NO in the photocatalyst. The transformation pathways and reaction mechanism of photocatalytic NO oxidation on CN and OH/Na co-functionalized carbon nitride have also been explicated by ESR spectroscopy and in situ DRIFTS and compared. This work provides a new method for simultaneously controlling the random transfer of carriers and adjusting the energy band structure of CN to optimize its photocatalytic efficiency. It is also possible to extend this strategy to improve the performance of other 2D layered catalysts for photocatalytic oxidation.


 Please wait while we load your content...
Please wait while we load your content...