Construction of 2D/2D Ni2P/CdS heterojunctions with significantly enhanced photocatalytic H2 evolution performance†
Abstract
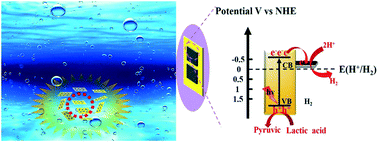
Two-dimensional (2D) materials have received tremendous attention in photocatalysis owing to their superior physicochemical properties including high specific surface areas, efficient interfacial charge separation and abundant surface active sites. Herein, 2D/2D Ni2P/CdS heterojunctions with Ni2P nanosheets (NSs) loaded on ultrathin CdS NSs were fabricated by combining liquid exfoliation of bulk CdS with a post-annealing procedure. The crystal phases, morphologies, microstructures, optical properties and specific surface areas of the as-prepared photocatalysts were thoroughly investigated by multiple detection techniques. The results of photocatalytic H2 evolution performance under visible light revealed that the optimal 2D/2D Ni2P/CdS heterojunction showed a significantly enhanced photocatalytic H2 evolution rate of 17.95 mmol g−1 h−1 using lactic acid as a sacrificial reagent, which was approximately 6 times higher than that of bare exfoliated CdS NSs, corresponding to the apparent quantum efficiency (AQE) of 4.2% under 420 nm monochromatic light. In addition, based on the analysis of electrochemical impedance spectroscopy (EIS), photocurrent response, steady photoluminescence (PL) spectroscopy and time-resolved photoluminescence (TRPL) spectroscopy, the enhanced photocatalytic H2 evolution activity was attributed to the effective separation and transfer of photogenerated charge carriers and generation of sufficient active sites. This work provided valid evidence that the use of 2D/2D heterojunction photocatalysts would be a promising technique in water splitting for H2 generation.


 Please wait while we load your content...
Please wait while we load your content...