Hydrodeoxygenation of ethyl stearate over Re-promoted Ru/TiO2 catalysts: rate enhancement and selectivity control by the addition of Re†
Abstract
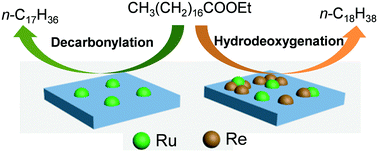
The hydrodeoxygenation of a model compound, ethyl stearate, of biomass-derived resources into long chain alkanes was investigated with Re-modified Ru/TiO2 catalysts. A monometallic Ru(1)/TiO2 catalyst, in which Ru was loaded in 1 wt%, gave a selectivity of >95% to the long chain alkanes of n-octadecane and n-heptadecane at almost 100% conversion at 220 °C, while another monometallic Re(10)/TiO2 catalyst was less active in spite of its higher metal loading (10 wt%). It is interesting that the addition of less active Re species to Ru(1)/TiO2 not only enhanced the rate of hydrodeoxygenation and increased the ratio of n-octadecane/n-heptadecane, while the high total selectivity towards alkanes remained unchanged. The alkane ratio depended on the content of Re added, increasing significantly from 0.5 for Ru(1)/TiO2 to 3.2 for Ru(1)Re(10)/TiO2. The impacts of Re addition on the catalytic performance of Ru/TiO2 were discussed on the basis of physicochemical properties characterized for Re-modified and unmodified Ru/TiO2 catalysts. It is suggested that the improved hydrodeoxygenation activity of Re-modified catalysts depends on the presence of Lewis acid sites, and the weak acid sites are important for determining catalytic selectivity. In addition, reaction pathways in the hydrodeoxygenation of ethyl stearate to n-octadecane and n-heptadecane were examined by control reactions with possible intermediates such as 1-octadecanol and stearic acid. The Re-modified Ru/TiO2 catalysts were shown to be recyclable during repeated runs when the same mole ratio of catalyst to substrate was used.


 Please wait while we load your content...
Please wait while we load your content...