Ultra-small subnano TiOx clusters as excellent cocatalysts for the photocatalytic degradation of tetracycline on plasmonic Ag/AgCl†
Abstract
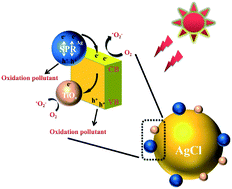
The separation efficiency and recombination rate of photogenerated electron–hole pairs play a vital role in photocatalytic activity. We report the incorporation of ultra-small subnano TiOx clusters as outstanding cocatalysts on the surface of plasmonic Ag/AgCl to boost the simulated sunlight photoreactivity. The formed structure is in favour of an efficient photoinduced electron–hole transfer process and suppressing the recombination rate, yielding an enhanced photocatalytic activity. The experimental results show that TiOx@Ag/AgCl exhibits an extraordinarily high activity in the photodegradation of tetracycline (TC) under simulated sunlight irradiation, which is about 21.7 times higher than that of bare Ag/AgCl. Their corresponding apparent quantum efficiency (AQE) is up to 0.917% at 435 nm and 17.459% at 350 nm during the photodegradation of TC, respectively. Moreover, TiOx@Ag/AgCl shows an outstanding long-term stability without obvious performance loss even after ten cycles of photodegradation. The possible enhancement mechanism of the reaction rate is proposed based on the compositional, structural, and optical property analysis. We found that h+ was the major reactive species in the photodegradation process of TC. Our findings on ultra-small TiOx cocatalysts would shed light on the design and development of highly efficient oxide cluster cocatalysts.


 Please wait while we load your content...
Please wait while we load your content...