Sb-doped polymeric carbon nitride with charge-capture centers for efficient charge separation and photocatalytic performance in H2 evolution and environmental remediation†
Abstract
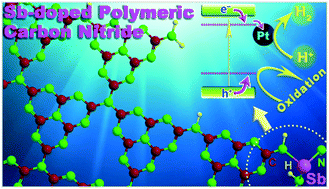
The high recombination efficiency of photogenerated charge carriers is the primary factor suppressing the photocatalytic performance of polymeric carbon nitride (CN). To enhance the charge separation, Sb-doped CN (CN/Sb) was firstly synthesized using antimonate as a doping and oxidizing agent. The N–SbIII bond was fabricated in CN/Sb via a redox process, which works as a charge-capture center, evidenced by experimental and theoretical calculation methods, and leads to a prominent enhancement in the charge separation and transfer performance of CN/Sb. CN/Sb exhibits much higher photocatalytic activity than CN in water splitting and environmental remediation. The visible-light photocatalytic hydrogen evolution rate of CN/Sb can reach ∼1.75 mmol g−1 h−1, which is ∼4.5 times that of CN, with an apparent quantum yield of 6.4% at 420 nm. Reactive oxygen species generation rates of CN/Sb are also much higher than those of CN. This work provides not only a new effective way for CN modification but also a deep insight into the metal-doping structure in the CN framework, which can guide future research on metal-doped CN photocatalysts.


 Please wait while we load your content...
Please wait while we load your content...