The elusive photocatalytic water splitting reaction using sunlight on suspended nanoparticles: is there a way forward?
Abstract
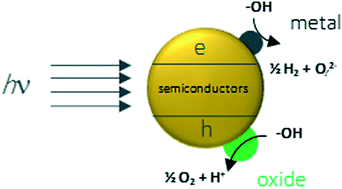
For many decades hydrogen production by photocatalytic methods has been pursued over a variety of semiconductors with probably over a thousand formulations of powder catalysts in many structures and compositions. Yet, with the exception of a few reports, water splitting to molecular hydrogen and oxygen has remained elusive. The only reproducible results are those involving other additives to water: electron donors or acceptors yielding either hydrogen or oxygen, but not both. The consequence of this is a system unrelated to water splitting but simply driven by the organic or inorganic redox potential. One may argue that thermodynamic limitations indicate that an inorganic semiconductor with a band gap within the spectrum of sunlight, and that is stable in water, cannot split water. Otherwise, it would not have existed on earth.
- This article is part of the themed collection: 2020 Catalysis Science & Technology Hot Articles


 Please wait while we load your content...
Please wait while we load your content...